Todd Hollon
Collaborative Neuro-Oncology
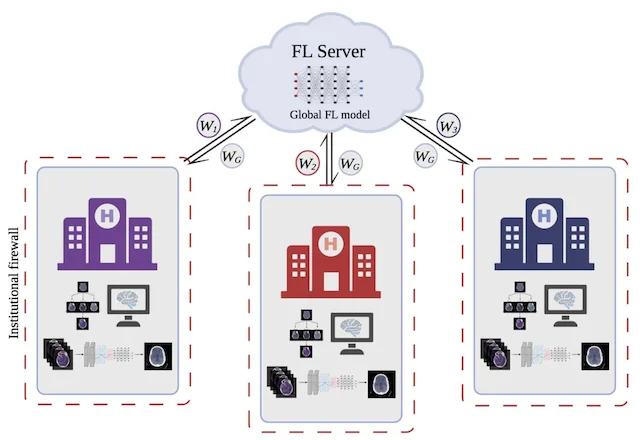
Progress against malignant brain tumors depends on team science: shared biobanks and registries, multi-disciplinary clinical trials, and partnerships across neurosurgery, neuro-oncology, pathology, and engineering. The MLiNS lab contributes to this ecosystem through translational studies that combine prospective trial design, rich tumor biology, and rigorous data science, including work on privacy-preserving collaboration (for example, federated learning across sites) so institutions can learn together without centralizing sensitive patient data. The publications below highlight representative collaborative neuro-oncology efforts.
Related publications
-
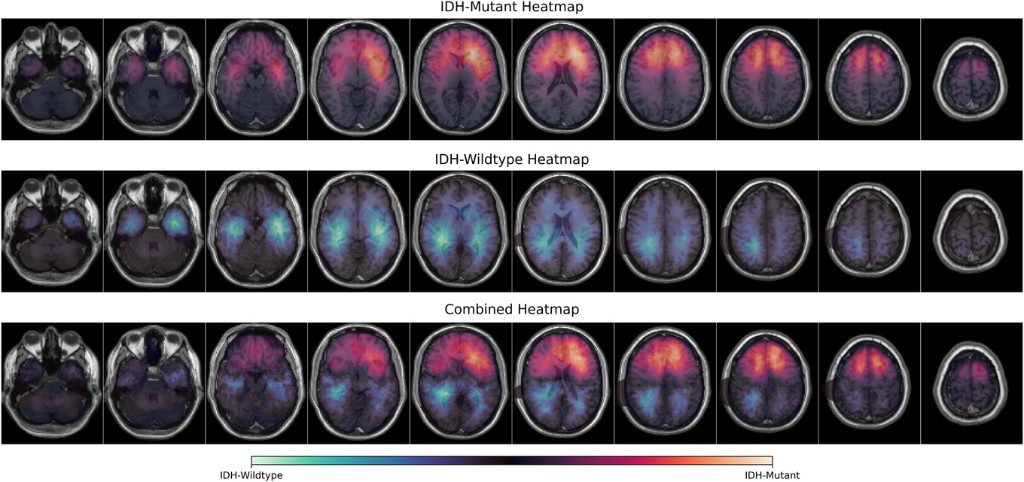
 NEUROSURGERY · 2026
NEUROSURGERY · 2026A multi-institutional spatial analysis characterizing where IDH-mutant gliomas arise in the brain, informing surgical planning, sampling, and interpretation of tumor biology across centers.
-
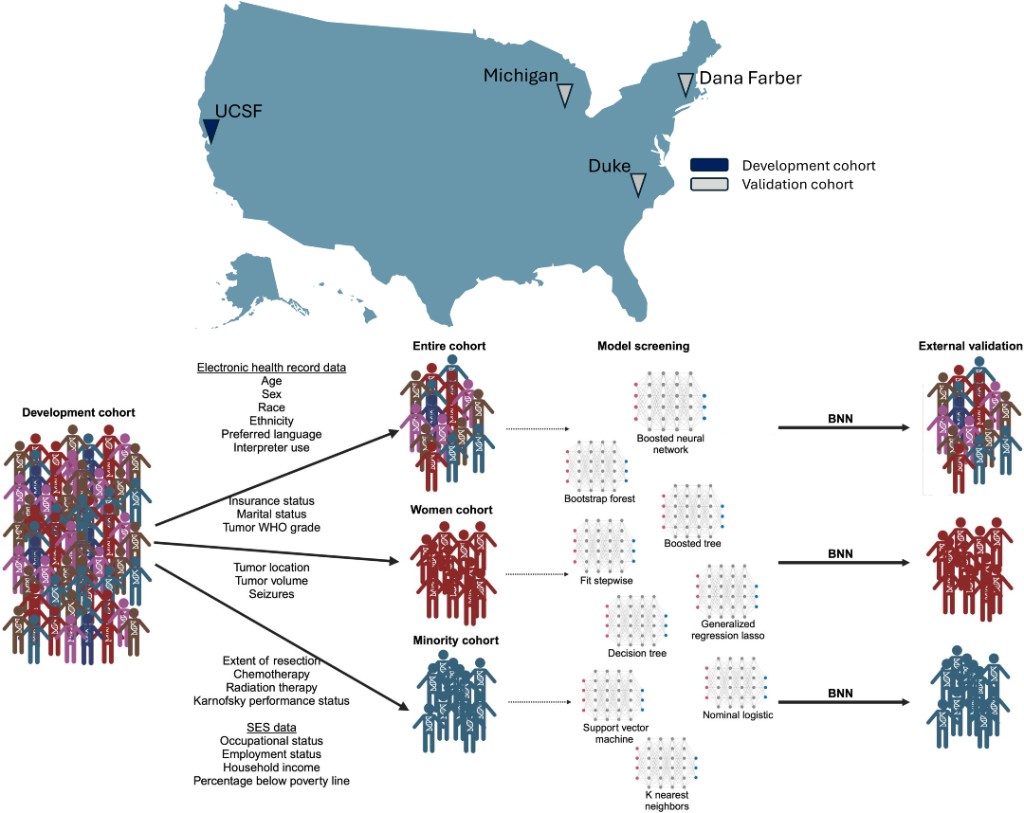
 SCIENCE ADVANCES · 2025
SCIENCE ADVANCES · 2025Supervised machine learning models predict clinical trial enrollment for adults with low- and high-grade glioma from routine data, with the goal of improving accrual and matching patients to studies.
-
 NATURE BIOMEDICAL ENGINEERING · 2024
NATURE BIOMEDICAL ENGINEERING · 2024Paired stimulated Raman histology and fluorescence microscopy map protoporphyrin IX (PpIX) during glioma resection, linking 5-ALA fluorescence to tissue context for more interpretable intraoperative guidance.
-
 The Lancet Oncology · 2023
The Lancet Oncology · 2023A phase 1, first-in-human trial of combined cytotoxic and immune-stimulatory gene therapy delivered to the resection cavity in adults with primary high-grade glioma, reporting safety and early efficacy signals.
-
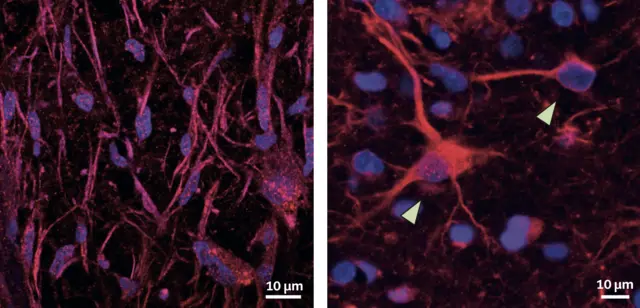
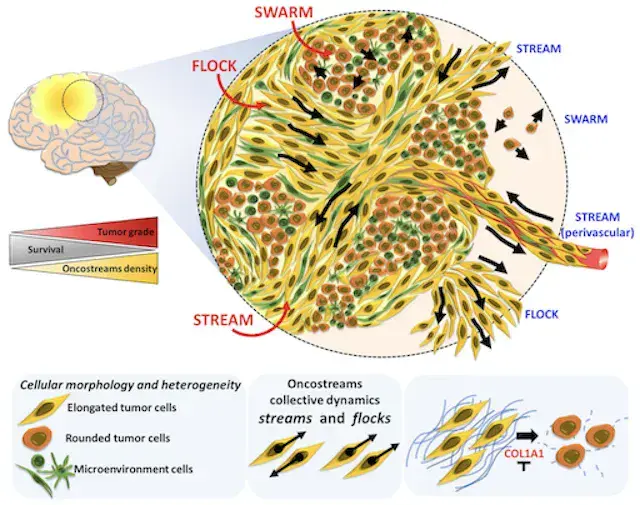
 NATURE COMMUNICATIONS · 2022
NATURE COMMUNICATIONS · 2022Spatiotemporal profiling of glioma heterogeneity implicates COL1A1 in the tumor microenvironment as a tractable target to slow progression, connecting matrix biology to therapeutic opportunity. The MLiNS lab contributed a semantic segmentation model to detect oncostreams in brain tumor microscopy images, supporting quantitative analysis of tumor architecture.
-
 NEUROSURGERY · 2022
NEUROSURGERY · 2022A practical overview of federated learning for multi-site clinical research in neurosurgery: how models can be trained collaboratively without centralizing raw patient data, and what that means for governance and discovery.